Atoms and Electrons Electronics Reference
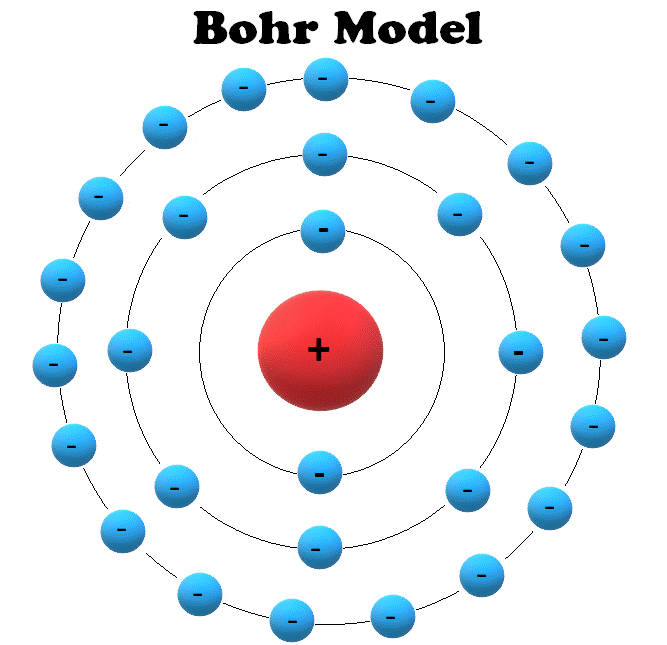
How does Niels Bohr's atomic model work? An overview of Niels Bohr's refinement of the Rutherford model. See all videos for this article Bohr model of the atom In the Bohr model of the atom, electrons travel in defined circular orbits around the nucleus. The orbits are labeled by an integer, the quantum number n.
Bohr Atom Modeli
1 Ry = e4me 8ϵ2 0h2 = 2.18 × 10 − 18 J. and this simplifies the allowed energies predicted by the Bohr model (Equation 7.4.11) as. En = − (2.18 × 10 − 18)Z2 n2 J = − Z2 n2 Ry. Hence, the energy of the electron in an atom also is quantized. Equation 7.4.12 gives the energies of the electronic states of the hydrogen atom.

konuşkan Sıçrama karar bohr modeli
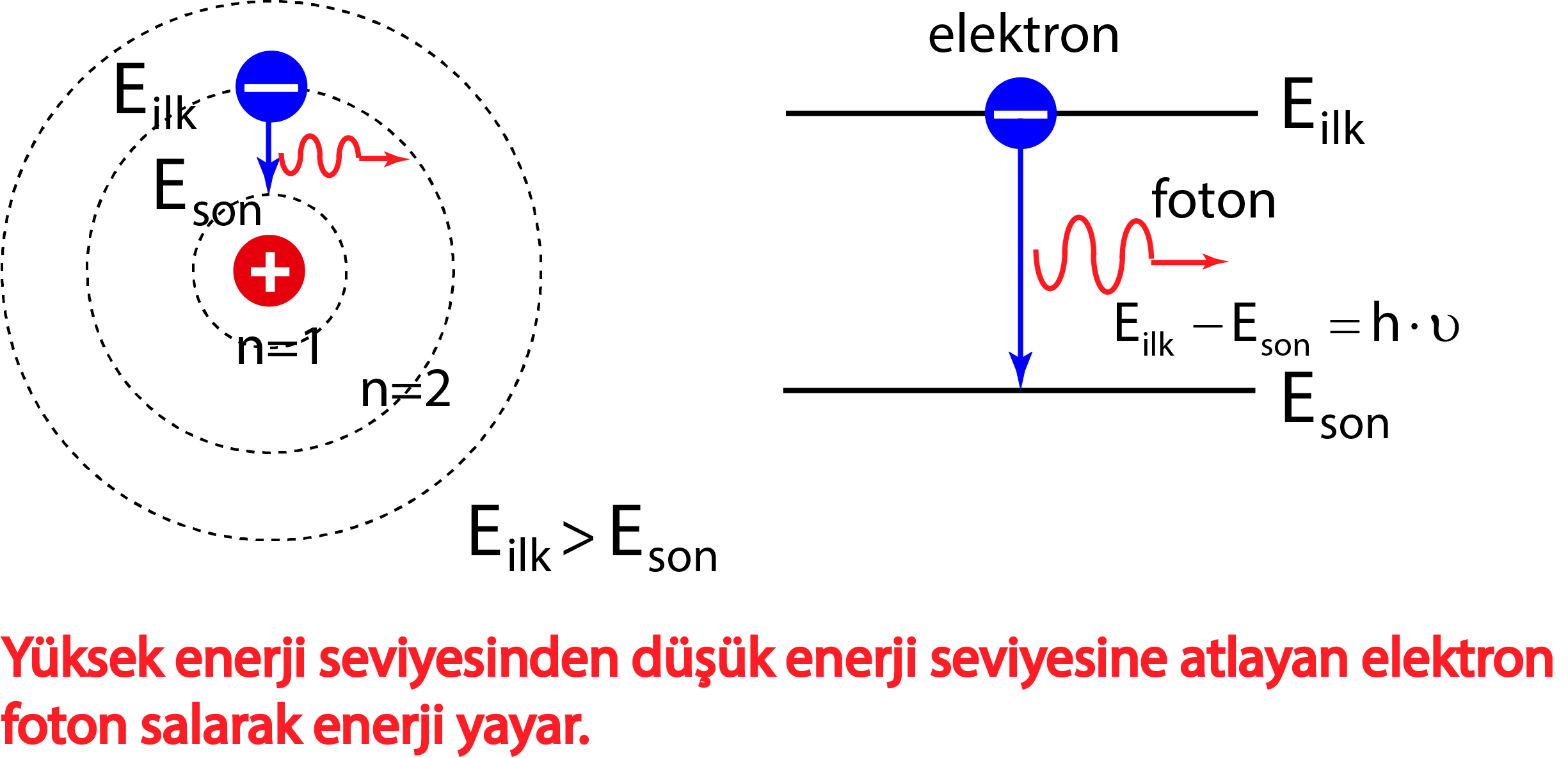
Bohr's model calculated the following energies for an electron in the shell, n. . : E ( n) = − 1 n 2 ⋅ 13.6 eV. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. h ν = Δ E = ( 1 n l o w 2 − 1 n h i g h 2) ⋅ 13.6 eV.

Atom Modeli ( Bohr Atom Modeli ) YouTube
Resources. Lecture Slides (PDF - 9.3MB) Periodic Table and Table of Constants. Lecture Summary. Prof. Sadoway talks about the principles of modern chemistry and how that led to the understanding of the structure of the atom.He details Bohr's postulates for the hydrogen atom and discusses how the Planck-Einstein relationship applies to electron transitions.
:max_bytes(150000):strip_icc()/atomic-structure-conceptual-artwork-99312661-58af58c75f9b5860467ff472.jpg)
History of Atomic Theory
Strafor köpük çöp şiş bakır tel ve alüminyum folyo kullanarak Bohr lityum Atom Modeli oluşturduk
mühendislik üçlü Titreme dalton atom modeli şekli
The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory.

Bohr's model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals. Main Idea A visualization of the Bohr model and the hydrogen spectrum. The Bohr model of the atom was proposed by Niels Bohr in 1913 as an expansion on and correction of the Rutherford model.
Bohr Model Atomic Theory Model Atomic Free Electron Model, PNG, 1067x1024px, Bohr Model, Area
Bohr's Atomic Model. Following the discoveries of hydrogen emission spectra and the photoelectric effect, the Danish physicist Niels Bohr (1885-1962) proposed a new model of the atom in 1915. Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist in states of constant energy that he called stationary states.

Bohr's Atomic Model
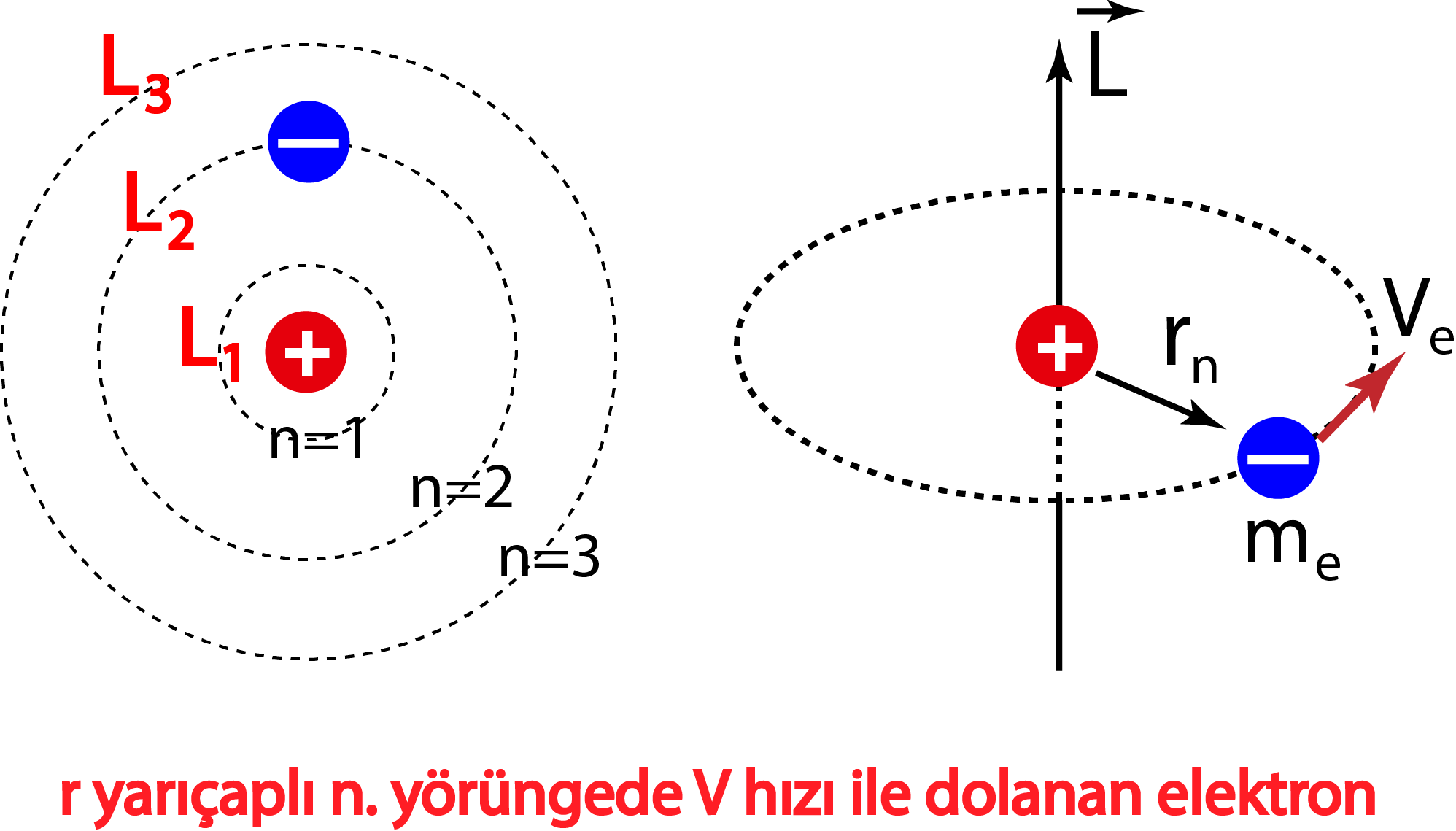
In 1913, Danish physicist Niels Bohr applied Max Planck's quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ().Note that Bohr stated that electrons in the atom follow elliptical orbits (not circles as is often pictured).
Bohr Atom Modeli Fizik. Net. Tr
Video \(\PageIndex{1}\): An introduction to the Bohr Model of the Atom. In 1913, Niels Bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism's prediction that the orbiting electron in hydrogen would continuously emit light. Instead, he incorporated into the classical mechanics description of the atom Planck's ideas of quantization and Einstein's finding that.

Atom Modeli Lityum ( Bohr Atom Modeli ) YouTube
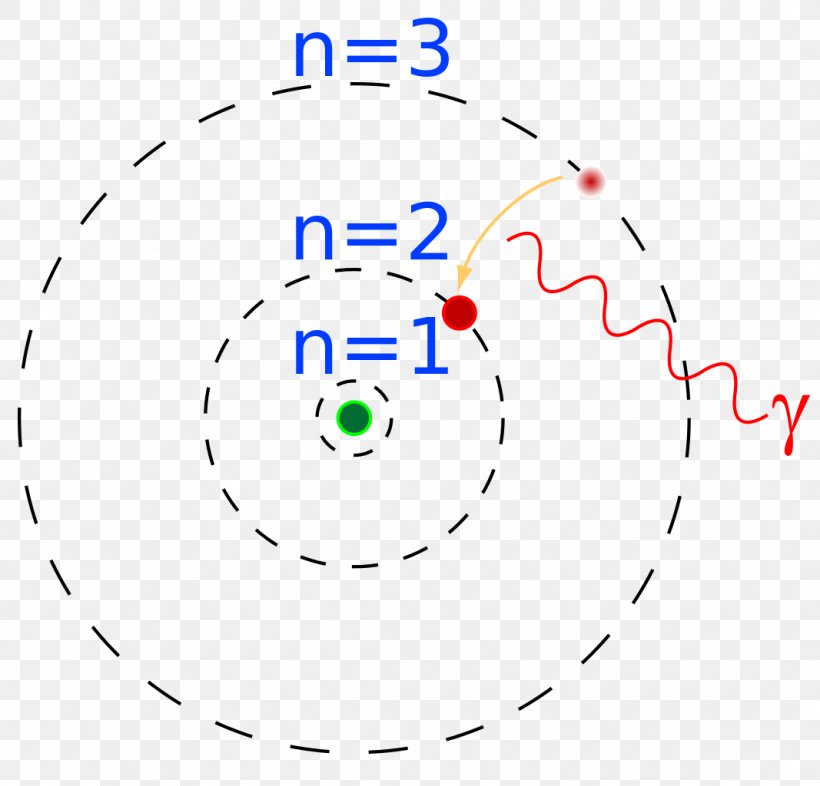
The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another. Only certain electron orbits are permitted.

Atom Modelleri(Dalton, thomson, rutherford, bohr atom modelleri) YouTube
🎈 12. Sınıf Konuları Serisinin Kitabını Sipariş Etmek İçin : https://bit.ly/3xb9oqW🔥 Bu Serinin Oynatma Listesi İçin : https://bit.ly/3B7HFIS🚀 TYT.

Oxygen molecule Atom model project, Science projects for kids, Atom model
ATOM MODELİ YAPILIŞI.Basit Atom Modeli yapımı.Atom modeli nasıl yapılır?Rutherford,Bohr.

ATOM MODELİ YAPILIŞI.Basit Atom Modeli yapımı.Atom modeli nasıl yapılır?Rutherford,Bohr. YouTube
A model of a helium atom shows a black circle which fades to white moving from the center to the outside. At the center of the circle is a tiny nucleus, consisting of two red circles and two purple circles.. A Bohr model of a chlorine atom shows a nucleus surrounded by three concentric rings. The ring closest to the nucleus is labelled n=1.

Aluminum Bohr Model Diagram, Using The Main Group Elements Of The Periodic Table To Draw Bohr
Rutherford explained the nucleus of an atom and Bohr modified that model into electrons and their energy levels. Bohr's model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. Bohr found that an electron located away from the nucleus has more energy, and the electron which.

Atom Modeli YouTube
Bohr Atom Modoli Modern Atom Teorisi (Bulut Modeli) Şimdi bu modelleri sıraysıyla görelim. Bu bir reklamdır: Dalton Atom Modeli (1803) John Dalton John Dalton (1766-1844) yaptığı çalışmalar sonucunda atomu şöyle tarif etmiştir: Maddeler atomlardan oluşur. (Doğru)